8 (4) A, C 0.1 gm of organic compound was analysed by Kjeldahl's method. In analysis produced NH, absorbed in 30 ml N/5 H,SO. The remaining acid required 20 ml N/10 NaOH
Por um escritor misterioso
Descrição
Click here:point_up_2:to get an answer to your question :writing_hand:84 a c01 gm of organic compound was analysed bykjeldahls method in analysis produced nhabsorbed

NR. Ammonia obtained from 0.4 g of an organic substance by Kjeldahl's method was absorbed in 30 ml. of 0-25 MH SO. The excess of the acid was neutralized by the addition
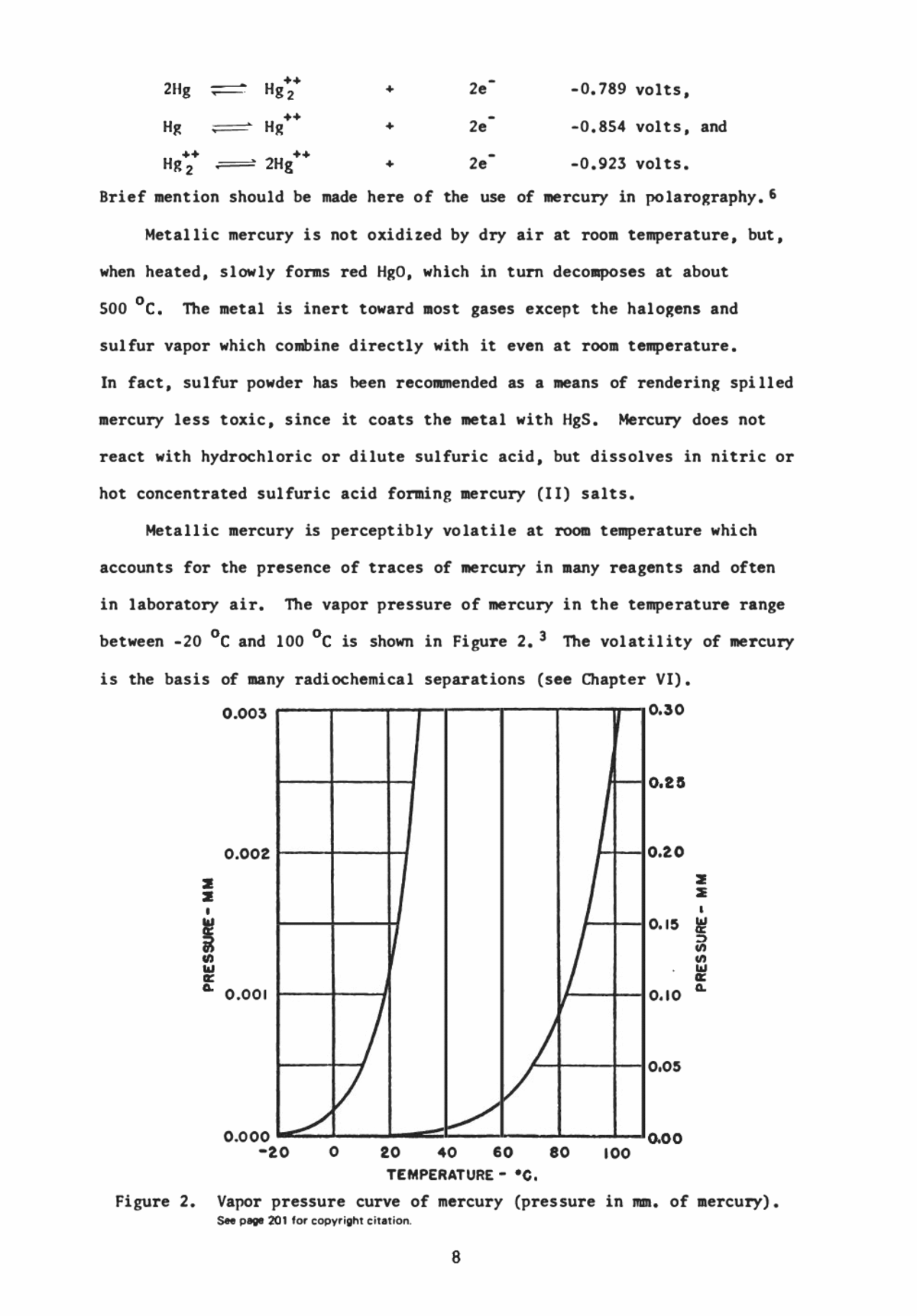
REVIEW OF FEATURES OF MERCURY CHEMISTRY OF CHIEF INTEREST TO RADIOCHEMISTS, Radiochemistry of Mercury

0.2 gm of an organic compound was analysed by kjeldahl's method the am

A sample of 0.50 g of an organic compound was treated according to Kjeldahl's method. The ammonia evolved was absorbed in 50 ml of 0.5 M ${{H }_{2}}$S${{O}_{4}}$. The residual acid required 60

An organic compound on analysis gave C=48gm,H=8gm and N =56gm. Volume of ..

1 0.1 g of organic compound in Kjeldahl method released ammonia gas which exactly neutralised 2 mL of 0.5 M sulphuric acid. Find percentage of nitrogen. (28 %)

PDF) Learners Guide to Soil Analysis

Psychrophilic and psychrotrophic clostridia: sporulation and

Methods for chemical analysis of soils - N.Z. Soil Bureau Scientific Reports - Manaaki Whenua Landcare Research Digital Library
18 29.5 mg of an organic compound containing nitrogen was digested according to Kjeldahls method and the evolved ammonia was absorbed in 20 mL of 0.1 M HCl solution. The excess of
Foods, Free Full-Text
53 Ammonia obtained from 0.4 g of an organic compound by kjeldahl's method was absorbed in 30 ml of 0.25 M H2So4 . The excess acid was neutralized by 30 ml of
de
por adulto (o preço varia de acordo com o tamanho do grupo)







